Immunogenicity
Immunogenicity describes the ability of a substance to trigger an immune response.
This can be:
Potential consequences of unwanted immunogenicity include:
Immunotoxicity
Immunotoxicity refers to harmful effects on the immune system caused by a therapeutic compound.
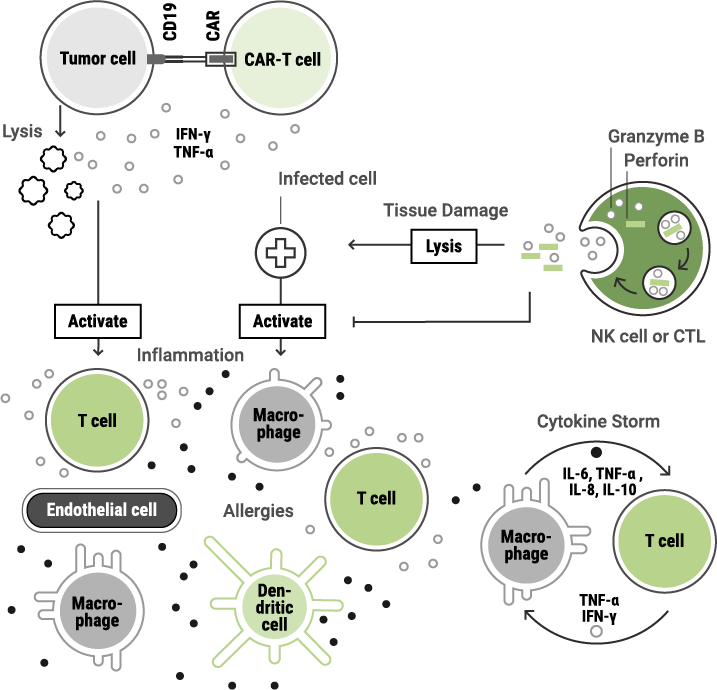
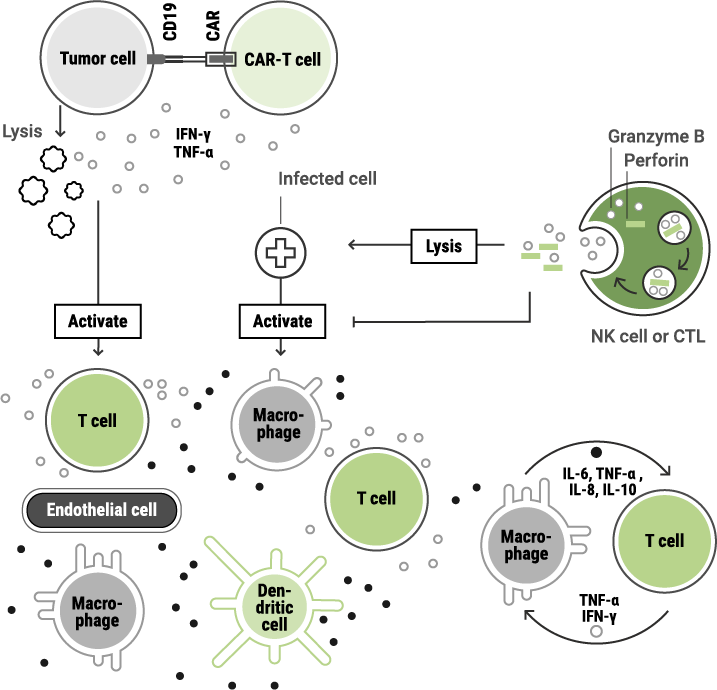
These effects may include:
Immunotoxic effects can occur even when the intended target is correctly addressed, making thorough evaluation essential.
Application areas
Assessment of immunogenicity and immunotoxicity is particularly relevant for:
At SYMBIOSIS, we focus on providing human-relevant biological materials that enable robust immune safety testing. Our resources support translationally relevant immune response assessment, population-representative testing strategies, and can be flexibly integrated into existing assay platforms.
Human-derived cell resources
SYMBIOSIS offers high-quality human immune cell materials to support immunogenicity and immunotoxicity studies, including:
These materials support:
Bioassay-services
Our bioassay services provide robust testing solutions for both Research Use Only (RUO) applications and studies conducted under Good Laboratory Practice (GLP) conditions, enabling critical insights into cellular and molecular interactions.
Key assays offered:
Contact us

